-
Table of Contents
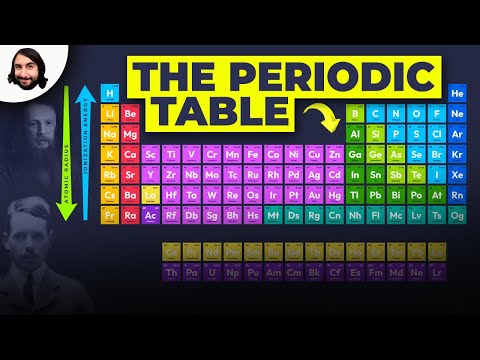
What Are the Trends in the Periodic Table
The periodic table is a fundamental tool in chemistry that organizes elements based on their atomic number, electron configuration, and recurring chemical properties.
. Understanding the trends within the periodic table can provide valuable insights into the behavior of elements and their interactions. In this article, we will explore some of the key trends in the periodic table and their significance.
1. Atomic Radius
One of the most prominent trends in the periodic table is the atomic radius, which refers to the size of an atom. As you move from left to right across a period in the periodic table, the atomic radius generally decreases. This is due to the increasing nuclear charge, which pulls the electrons closer to the nucleus. Conversely, as you move down a group, the atomic radius tends to increase. This is because the number of electron shells increases, leading to a larger atomic size.
2. Ionization Energy
Ionization energy is the energy required to remove an electron from an atom. In general, ionization energy increases as you move from left to right across a period in the periodic table. This is because the electrons are held more tightly due to the increasing nuclear charge. Conversely, ionization energy tends to decrease as you move down a group. This is because the outermost electrons are farther from the nucleus and experience less attraction.
3. Electronegativity
Electronegativity is a measure of an atom’s ability to attract and hold onto electrons. Similar to ionization energy, electronegativity tends to increase as you move from left to right across a period in the periodic table. This is because atoms with higher electronegativity values have a greater ability to attract electrons. Conversely, electronegativity tends to decrease as you move down a group.
4. Metallic and Non-Metallic Properties
Elements in the periodic table can be classified as metals, non-metals, or metalloids based on their properties. Metals tend to be located on the left side of the periodic table, while non-metals are found on the right side. Metalloids exhibit properties of both metals and non-metals and are located along the staircase line in the periodic table.
Summary
Understanding the trends in the periodic table is essential for predicting the behavior of elements and their compounds. By recognizing patterns such as atomic radius, ionization energy, electronegativity, and metallic properties, chemists can make informed decisions about reactions, bonding, and material properties. The periodic table serves as a roadmap for exploring the vast world of chemistry and provides a framework for organizing the building blocks of matter.